We conduct research in two major areas: Genome Editing and Plant Synthetic Biology.
-
 Precise Genome Editing: Mechanisms and Technologies
Precise Genome Editing: Mechanisms and TechnologiesPrecise genome editing enables the targeted rewriting of genomes. This technology is revolutionizing our ability to dissect gene function, reprogram biological processes, and accelerate crop improvement. Our lab investigates the molecular mechanisms underlying genome editing processes and utilizes these findings to develop new innovations for precision editing in plants. The key questions we explore include: How did genome-editing enzymes evolve in nature? Which natural biological elements possess programmable potential and can be harnessed as novel editing systems? What factors limit editing efficiency, specificity,and scalability? How do genome editing tools behave in different plant cellular contexts? Can we achieve high-precision editing without introducing foreign DNA?
On one hand, we investigate previously untapped biological elements fortheir potential as programmable genome editing systems. Through systematic mining of their functional diversity, structural features, and molecular evolutionary trajectories, we aim to uncover novel editing capacities and underlying mechanisms. This effort has led to the development of new genome editing systems with distinct modes of action, such as TranC (Transposon and CRISPR intermediate), which expand the phylogenetic and mechanistic boundaries of current genome editing toolkits. On the other hand, we have developed a versatile suite of editing technologies—including base editing, prime editing, and targeted large-chromosomal manipulation—which enables precise DNA substitutions, insertions, and deletions across a broad range of species, including rice, wheat, maize, soybean and more. By elucidating editing mechanisms and refining new editing tools, we are advancing genome editing as a core platform for engineering climate-resilient and high-yielding crops.
More -
 AI-Guided Tool Development
AI-Guided Tool DevelopmentArtificial intelligence (AI) is rapidly transforming the state of biotechnology, enabling genome editing tool development to become faster, smarter, and more iterative in both discovery and optimization processes. By integrating structural prediction, evolutionary modeling, and experimental feedback, AI enables rational design far beyond the limits of lab-based protein engineering approaches.
In 2023, we introduced a pioneering AI-driven structural clustering method for enzyme mining, which, for the first time, introduced the concept of using parallel high-throughput AI to expedite genome editor discovery. Building on this, we established a closed-loop AI-assisted platform for enzyme optimization, accelerating the evolution of genome editing tools. These innovations are pushing the boundaries of programmable biology, with broad implications for plant biotechnology, synthetic biology, and precision medicine.
More -
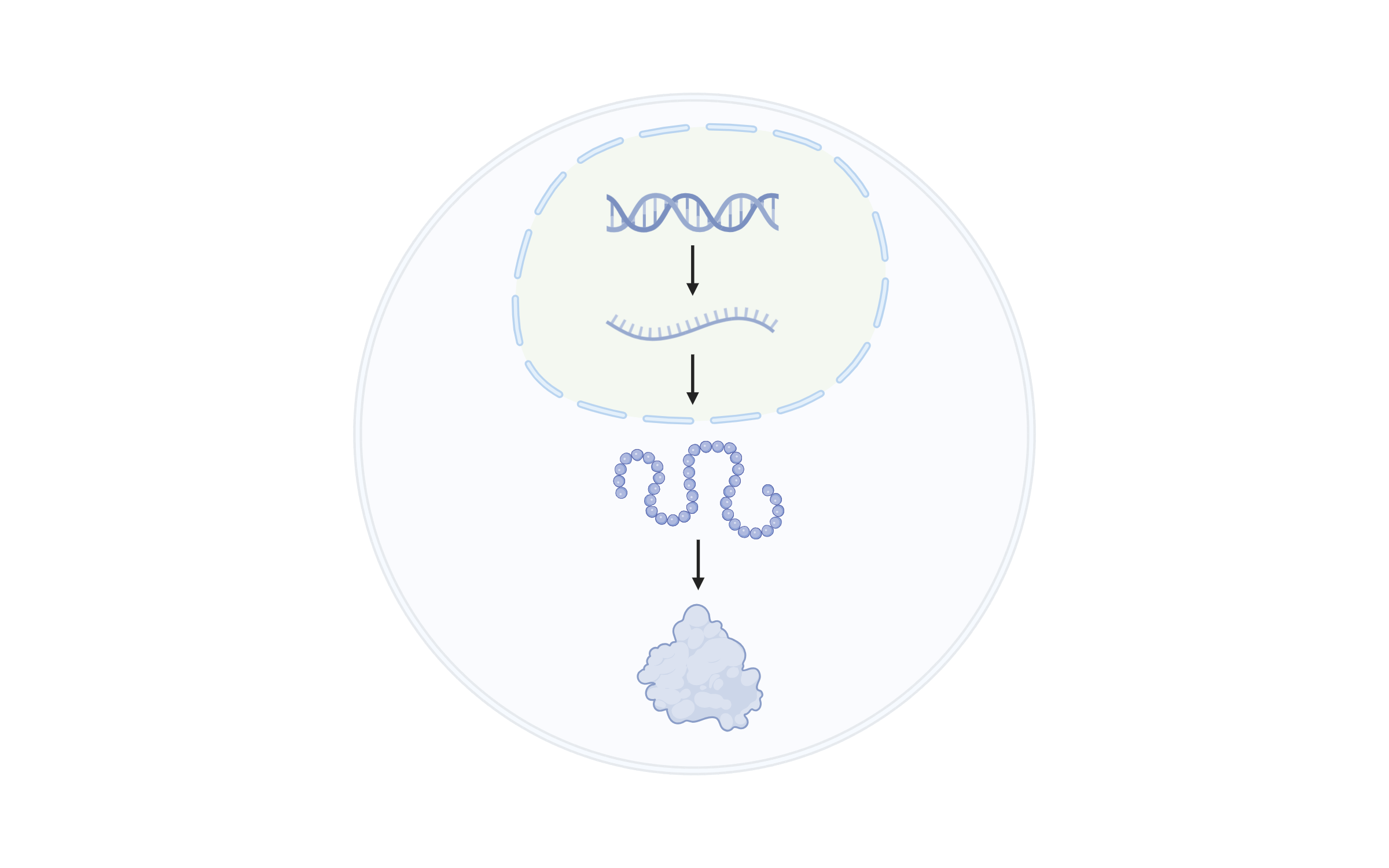 Programmable Gene Regulation
Programmable Gene RegulationPrecise control of gene expression is essential for engineering complex traits in crops. Our lab develops programmable regulatory strategies at multiple levels—transcriptional, post-transcriptional, and translational—to achieve quantitative tuning of gene expression in plants.
At the translational level, we reprogram upstream open reading frames (uORFs) to fine-tune protein translation. At the post-transcriptional level, we modulate pre-mRNA alternative splicing by targeting conserved splice sites. These tools have enabled customized trait design in lettuce, strawberry, and rice, improving nutritional content, sugar accumulation, plant architecture, and more.
Our continuing efforts seek to advance CRISPR-based large-fragment engineering, RNA splicing control, translational fine-tuning, and nuclear architecture manipulation. By combining genome editing, directed evolution, single-cell multi-omics, and machine learning, we aim to decode regulatory networks and establish a framework for multiscale, programmable gene expression in plants.
More -
 Directed Evolution in Plants
Directed Evolution in PlantsDirected evolution is a powerful strategy for engineering protein function, yet scalable systems in plants remain limited. To address this, we established the Geminivirus Replicon-Assisted in Planta Evolution (GRAPE) platform, which harnesses geminivirus-based rolling circle replication for rapid mutagenesis and functional selection in plants.
GRAPE enables the screening of up to 10⁵ variants on a single tobacco leafin just four days. Using this system, we have evolved a suppressor tRNA for efficient stop codon readthrough, an enhanced NRC3 immune receptor for resistance to the pathogen effector SS15, and a Pikm-1 variant with an expanded recognition spectrum. We are continuing to upgrade GRAPE to into an advanced, modular, high-throughput platform for evolving plant proteins and regulatory elements to promote sustainable agriculture and crop resilience.
More -
 Plant Regeneration and Delivery Systems
Plant Regeneration and Delivery SystemsThe development of efficient delivery and regeneration systems remains a critical bottleneck for broad applications of plant genome editing. Our lab develops innovative systems to overcome genotype dependence, long regeneration cycles, and species recalcitrance during these editing processes.
We have established robust delivery platforms for wheat, including the TaGRF4–TaGIF1-based co-transformation approach and BSMV-sgRNA viral vectors, enabling heritable editing across diverse wheat cultivars. In parallel, we developed a DNA-free RNP and mRNA delivery method that meets biosafety requirements. By integrating single-cell transcriptomics, metabolomics, and proteomics, we have identified key regeneration factors that enable transformation and plant regeneration in recalcitrant species such as indica rice, tetraploid wild rice, and octoploid strawberry.
Moving forward, we aim to optimize conventional transformation methods such as Agrobacterium-mediated delivery and biolistics, and to extend DNA-free editing to horticultural crops and forest trees. By combining AI-assisted regulatory network modeling, directed evolution of regeneration factors, and high-throughput screening, we are building a programmable framework for efficient and genotype-independent plant regeneration.
More -
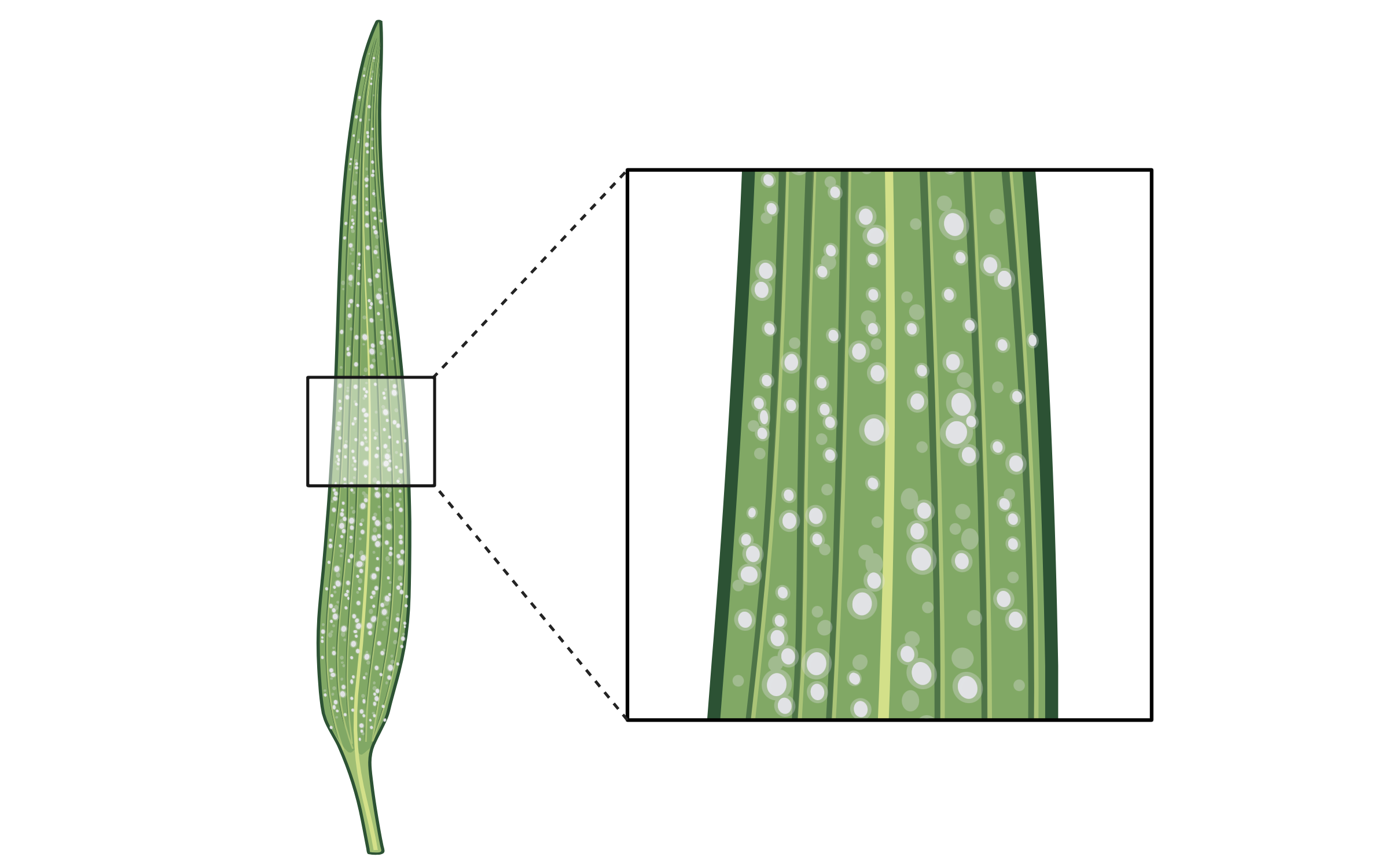 Crop Engineering and Trait Innovation
Crop Engineering and Trait InnovationBiotechnology is fundamentally changing how we engineer crop traits, enabling the development of novel, high-value cultivars for food, health, and sustainability. Our lab is driving this transformation by integrating genome editing, multi-omics, and computational design into trait development and breeding pipelines.
We have pioneered approaches such as in planta saturation mutagenesis, de novo domestication of wild species, and precision engineering of complex traits—including targeted editing in polyploid crops. These approaches have enabled the generation of plants with improved disease resistance, enhanced nutrition, and consumer-desired traits such as extended shelf life and flavor profile changes.
Looking ahead, we are integrating artificial intelligence, high-throughput functional screening, and programmable editing platforms to build intelligent crop design pipelines. These efforts are bridging the gap between fundamental discoveries and real-world agricultural deployment, supporting the development of resilient, high-performance crops for future food systems.
More
